| Information | |
|---|---|
| Full Name: | Zeta-chain associated protein kinase |
| Abbreviation: | ZAP70 |
| Molecular Mass: | 70 kDa |
| OMIM: | 176947 |
| PDB: | 2OZO |
Introduction
Zeta-chain associated protein kinase 70 (ZAP-70) is a cytoplasmic protein of 70 kilo-Daltons. It is the first protein substrate of the T-cell receptor (TCR)/CD3 complex situated on the plasma membrane of T lymphocytes. Therefore, ZAP-70 is responsible for the initiation of all the different pathways in the T-cell signaling cascade, and plays a critical role in the development of cell-mediated immune response.
Molecular surface map of ZAP70
T-Cell Receptor Signalling Pathway
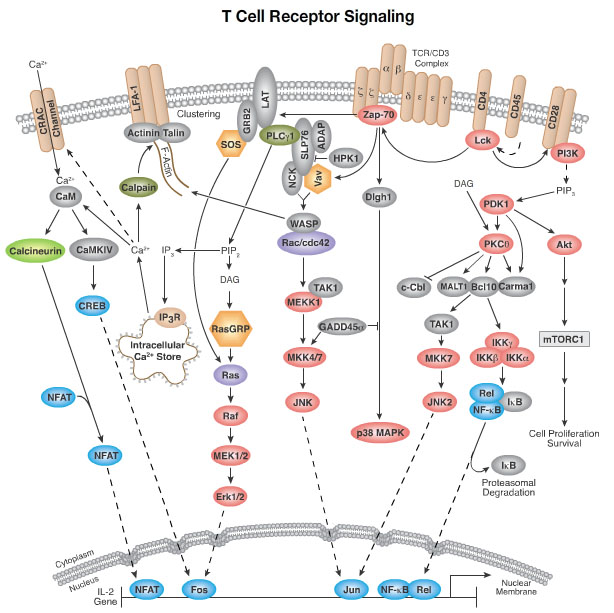
TCR is activated upon binding with the peptide-MHC (Major histocompatibility complex) complex. This interaction is enhanced via recruitment of co-receptors CD4 (or CD8) whose cytoplasmic tails are associated with a lymphocyte cell-specific protein tyrosine kinase (Lck). [1,2,3] Lck phosphorylates the immunoreceptor tyrosine-based activation motif (ITAM) in the CD3 zeta chain which is a special motif that consists of four amino acids with signature sequence YXXI/L. [1, 7,8] Once the ITAM is double phosphorylated, the affinity toward ZAP-70 greatly increases and allows ZAP-70 interaction. [1, 2, 3] These results in a conformational change in ZAP-70 from its autoinhibited form permitting the uncovering of the regulatory sites to be phosphorylated by Lck and thus activated. In addition, ZAP-70 also undergoes auto-phosphorylation in the tyrosine residues present in its activation loop of the kinase domain to increase its catalytic activity. [3, 7, 8] The activated ZAP-70 is now able to interact with its downstream molecules to perform cell signaling.
Brief Mechanism
The linker of active T-cells (LAT) and the SH2-domain-containing leukocyte protein (SLP-76) are two main substrates of ZAP-70. Both LAT and SLP-76 are scaffolding proteins (adaptor proteins) activated upon phosphorylation by ZAP-70. LAT activates growth factor receptor bound protein 2 (Grb-2). Grb-2 together with SOS help activates GTPase RAS. This pathway eventually enhanced FOS gene transcription. As for SLP-76, ZAP-70 phosphorylates SLP-76 at specific sites and therefore regulates SLP-76-Vav binding [9] for its final function of enhancing the transcription of the JUN genes. [1] SLP-76 is also able to increase FOS gene expression by interacting with Grb-2 and Cγ-1 through joining the Ras activation pathway. [4, 6] FOS and JUN dimer form gene transcriptional factor that involves cell proliferation and cell differentiation. [4, 5] In brief, the overall effect of ZAP-70 is a series of activities which lead to the eventual proliferation and differentiation of T cells and production of cytokines.
A more complete description with diagramsDisease Links
The most well-known diseases implicated in ZAP-70 are Severe Combined Immunodeficiency (SCID) and B-cell chronic lymphocytic leukemia (B-CLL) and both result from mutations causing dysfunction of ZAP-70. ZAP-70 is essential in the regulation of T-cell development as it was demonstrated in ZAP-70 deficient mice a failure in this process, where the allocation of which T cell will have either CD8+ or CD4+ (never both), so-called positive/negative selection, on its surface, was found to have this transition arrested. The presence of double positives usually leads to spontaneous apoptosis. Thus, when ZAP-70 is absent, T-Cell activation never takes place. ZAP-70 is also a good marker for sorting B-CLL into subgroups. However, whether ZAP-70 is involved in B-cell signaling pathways remain controversial, though no direct involvement has been found at present.
References
- M.C. Bene 2006. 'What is Azp-70?'- Cytometry Part B: Clinical Cytometry DOI 10.1002/cyto.b
- Cellcignall.com- on T cell receptor pathways
- Haopeng Wang, Theresa A. Kadlecek, Byron B. Au-Yeung, Hanna E. Sjölin Goodfellow, Lih-Yun Hsu, Tanya S. Freedman and Arthur Weiss. 2010: 'ZAP-70: An Essential Kinase in T-cell Signaling.'
- Weiguo Zhang, Joanne Sloan-Lancaster, Jason Kitchen, Ronald P Trible, Lawrence E Samelson. Cell, Vol.92, 1998: 'LAT: The ZAP-70 Tyrosine Kinase Substrate that Links T Cell Receptor to Cellular Activation'
- C Abate, L Patel, FJ Rauscher, 3rd and T Curran. Science, 1990. Vol.249 1157-1161 'Redox regulation of fos and jun DNA-binding activity in vitro'
- Juliane Bubeck Wardenburg, Chong Fu, Janet K. Jackman, Horst Flotow, Sandra E. Wilkinson, David H. Williams, Robin Johnson, Guanghui Kong, Andrew C. Chan and Paul R. Findell. 1996. The Journal of Biological Chemistry, 271, 29641-19644: 'Phosphorylation of SLP-76 by the ZAP-70 Protein-tyrosine Kinase Is Required for T-cell Receptor Function'
- Wang H, Kadlecek TA, Au-Yeung BB, Goodfellow HE, Hsu LY, Freedman TS, Weiss A. Cold Spring Harb Perspect Biol. 2010 May; 2(5):a002279. 'ZAP-70: an essential kinase in T-cell signaling.'
- Visco C, Magistrelli G, Bosotti R, Perego R, Rusconi L, Toma S, Zamai M, Acuto O, Isacchi A. Biochemistry. 2000 Mar 14; 39(10):2784-91. 'Activation of ZAP-70 tyrosine kinase due to a structural rearrangement induced by tyrosine phosphorylation and/or ITAM binding.'
- Raab M, da Silva AJ, Findell PR, Rudd CE. Immunity. 1997 Feb; 6(2):155-64. 'Regulation of Vav-SLP-76 binding by ZAP-70 and its relevance to TCR zeta/CD3 induction of interleukin-2.'