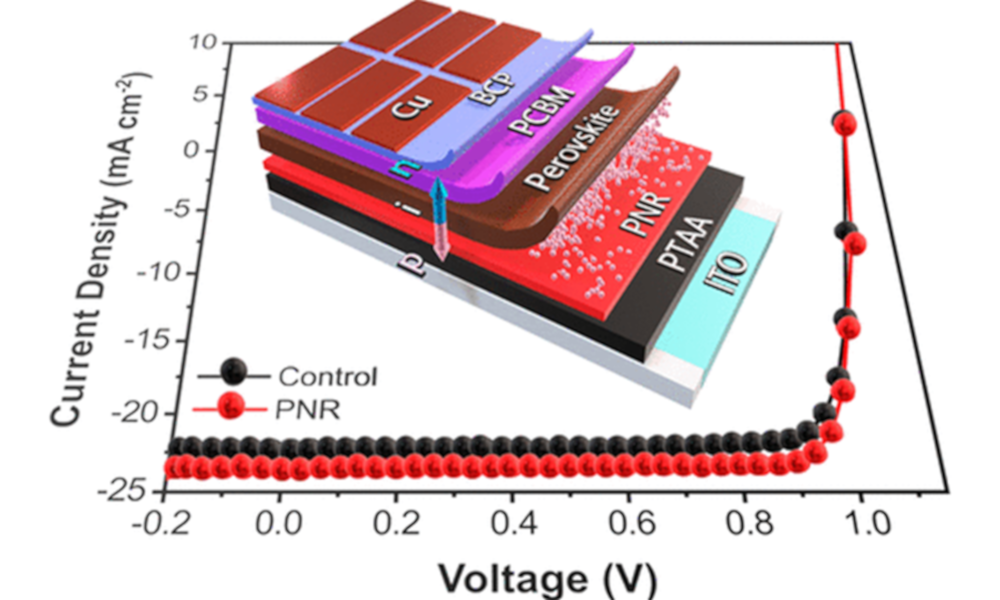
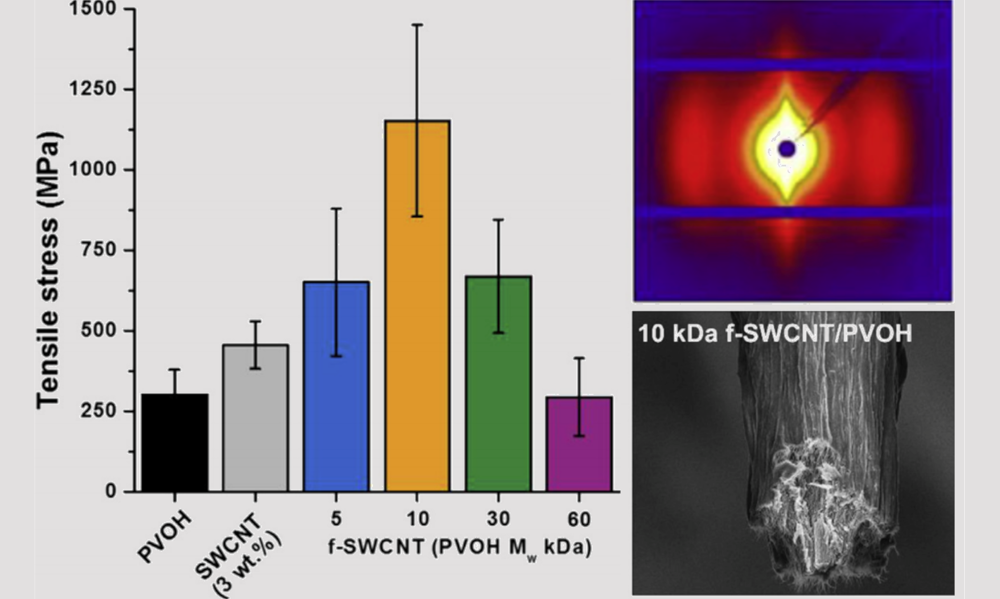
Spontaneous Dissolution of Nanomaterials
Harnessing the power of thermodynamics to avoid damage and driving scalability
Most liquid phase dispersions of nanomaterials are kinetically trapped by energy-matched solvents or coating of surfactants, having previously been ripped into smaller stacks/bundles by sonication or shear mixing. This application of shear force is not only inefficient in creating the individualised nanomaterials we want, but actively damages the nanomaterials.
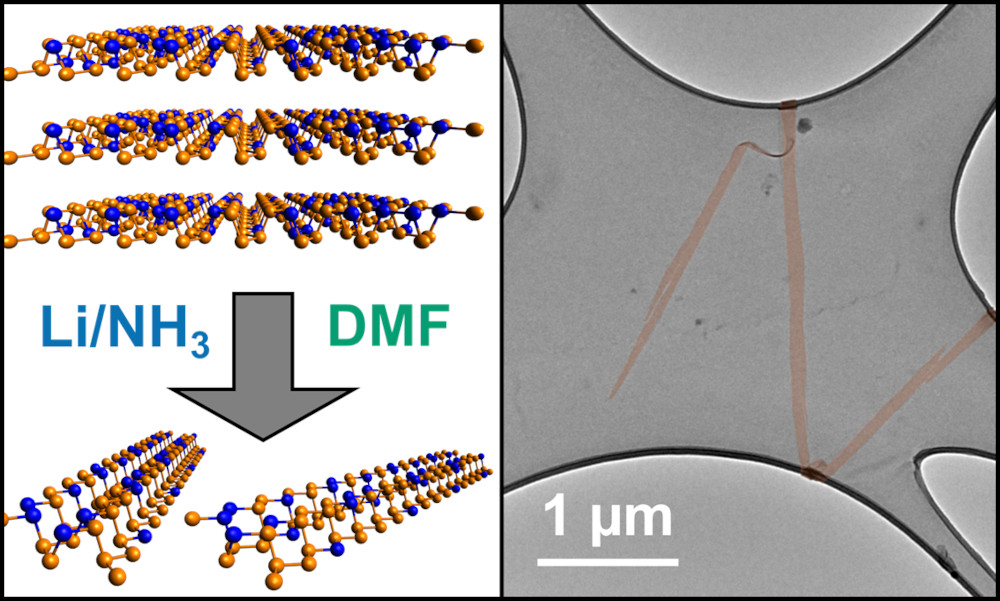
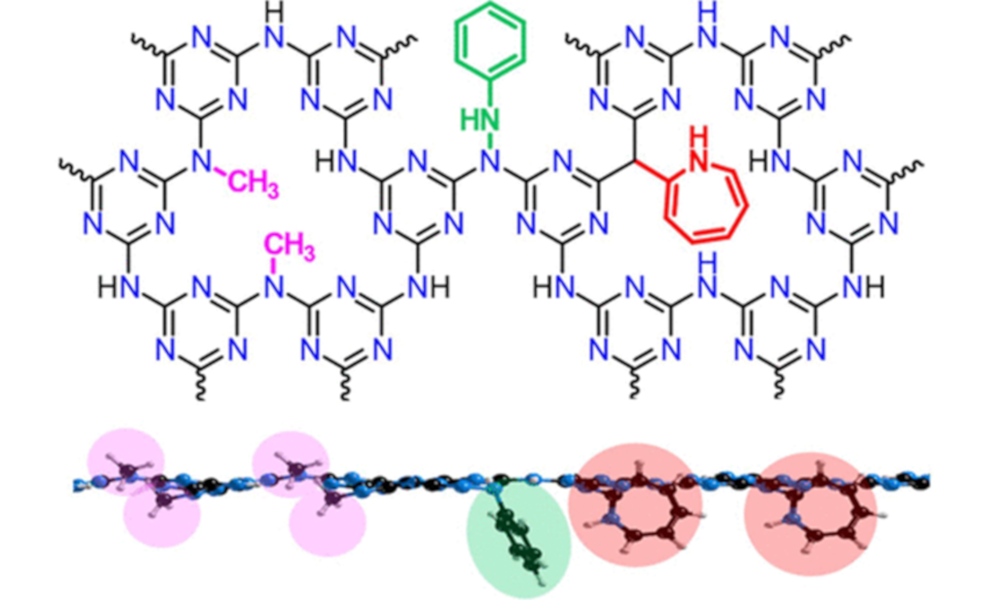
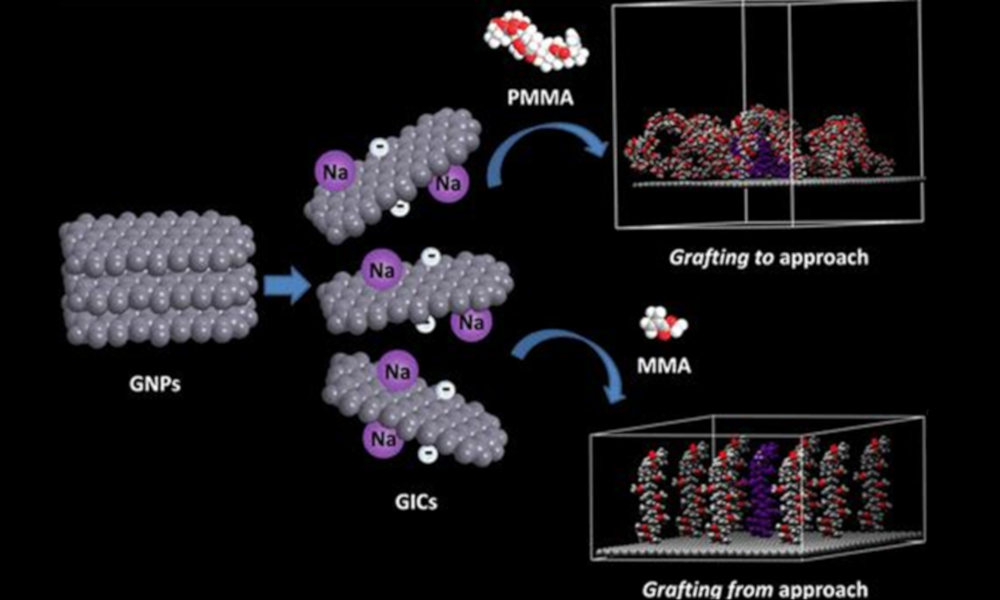
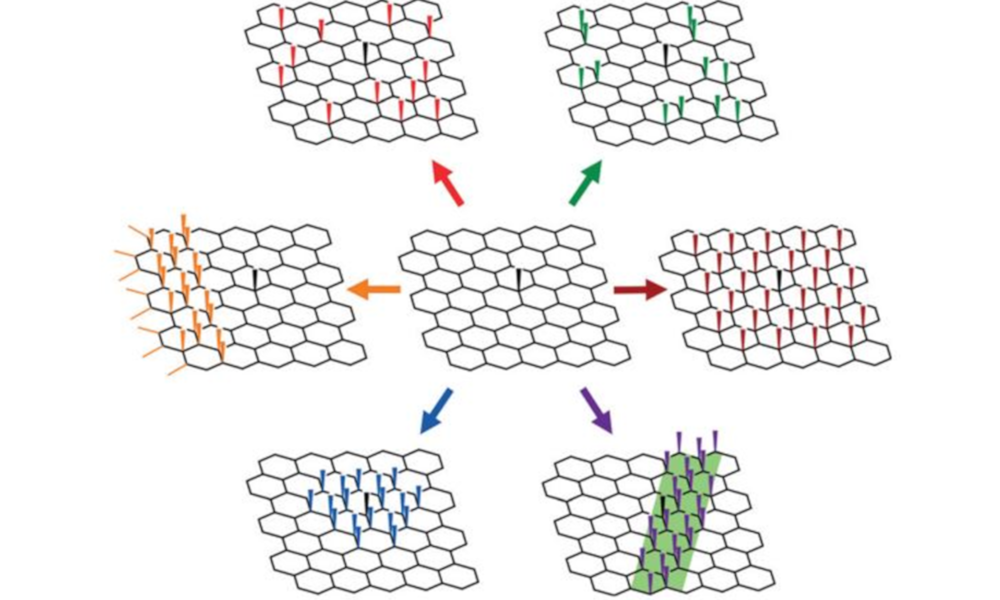
In contrast, we use spontaneous dissolution, where the ground state and thermodynamic product is the individual nanomaterial in solution. Now there is a driving force for effective exfoliation/exfibrillation, with the scalability limited by the nanomaterial synthesis and the size of the bucket you use for dissolution. The chemistries needed to invoke dissolution changes with the nanomaterial, but common approaches involve introducing charge onto the nanomaterial with superacids or group 1 metals.
These nanomaterial solutions are not only useful (and underpin much of the rest of our research), but pose interesting fundamental questions about the nature of solvation, as well as providing high concentration models for charged, nano-surfaces to probe a range of theories.