Sleep Disruption in Neurological Disease
Sleep disruption is a major aspect of many disorders, including neurodegenerative diseases like Alzheimer's and neurodevelopmental disorders like autism. The Rihel lab has several projects investigating how disease processes may affect sleep.
In one project, spearheaded by PhD student Francois Kroll, we are systematically testing whether genes associated with Alzheimer's Disease have effects on sleep or the circadian rhythm. He hypothesises that some of the mutations that increase risk of developing Alzheimer's at later ages might do so because they perturb sleep during the individual's lifetime. For this purpose, he has optimised a CRISPR-Cas9 gene editing method to quickly disrupt any gene in zebrafish, and is using this method to systematically test the behavioral effects of making mutations in genes associated with Alzheimer's Disease.
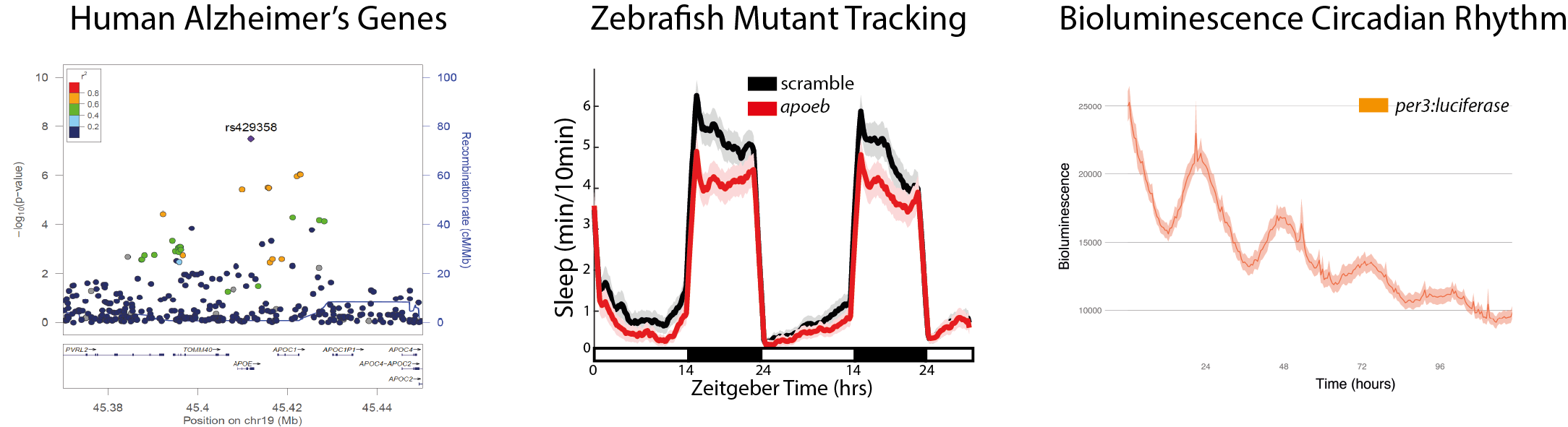
Some human genetic variants are associated with Alzheimer's Disease and changes in sleep (left). By making zebrafish mutants in these candidate genes (middle), we can test for changes in sleep behavior. We can also use a bioluminescence assay (right) to measure changes in the 24-hour circadian rhythm — these fish express the firefly protein luciferase under the control of a clock gene, causing them to glow with a rhythmic pattern.
Sleep and Alzheimer's Disease
Sleep and Alzheimer's Disease may be intimately linked because of variations in the production and clearance of amyloid beta and tau, the major protein components of the plaques and tangles that build up in the brains of Alzheimer's patients. These proteins are secreted from the brain predominantly during waking and are cleared from the brain during sleep. This has led to the suggestion that a vicious cycle may develop, in which damage from the buildup of amyloid beta and tau leads to disrupted sleep, which in turn allows more damaging molecules to accumulate, leading to further disrupted sleep.
The Rihel lab has been investigating the links between amyloid beta and sleep in zebrafish. One of the surprising findings has been that amyloid beta can either induce or suppress sleep, depending on the molecular conformation it forms. By making gene edits to candidate molecular targets, we are starting to uncover some of the pathways involved in linking Alzheimer's molecules to sleep-wake regulation.
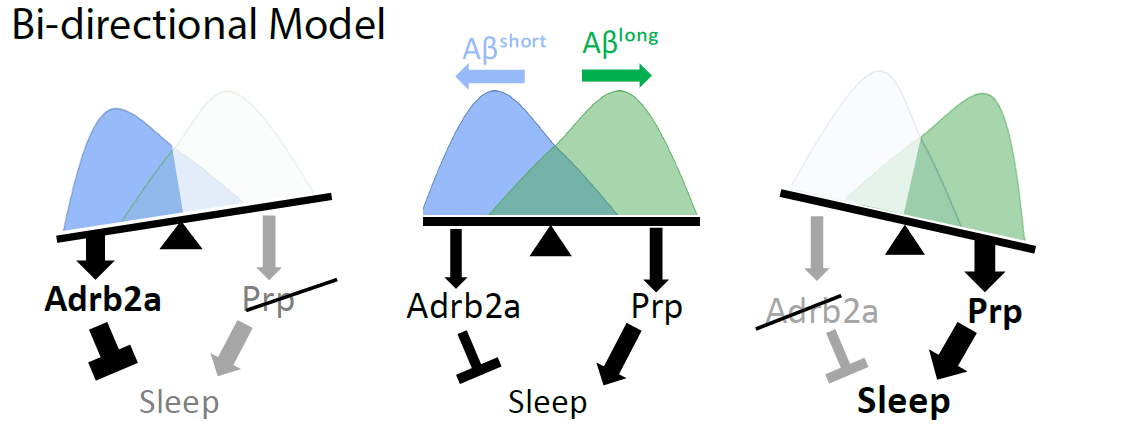
We have proposed a model in which short forms of Amyloid beta (blue) inhibit sleep and long forms (green) enhance sleep. When putative Amyloid beta binding partners are mutated, the see-saw tilts in favor of more or less sleep.
Sleep and Brain Clearance
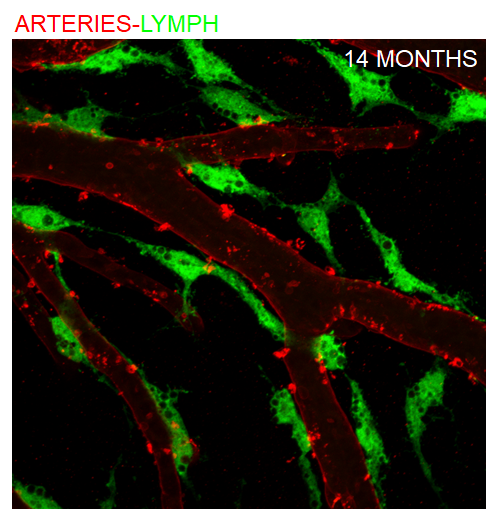
Brain-Lymphatic Endothelial Cells (BLECs) in the zebrafish meninges. These cells are capable of taking up macromolecules including amyloid beta and may play an important role in brain clearance.
The "Washing Machine" hypothesis proposes that during sleep, toxic by-products that build up during wakefulness are cleared from the brain, as the space between neurons expands and becomes more accessible to the surrounding cerebrospinal fluid. The Rihel lab has been investigating this phenomenon in zebrafish, in which we co-discovered a novel cell type residing in the meninges, the fibrous protective covering of the zebrafish brain.
More recently, we showed that these cells are also present in the mouse and human meninges. These cells, called Brain-Lymphatic Endothelial Cells (BLECs), are related to the lymphatic vessels that drain fluid from other parts of the body, although they do not normally form tubes. BLECs are capable of taking up macromolecules injected into the brain, including amyloid beta. Current projects are designed to observe how BLECs respond to changes in sleep/wake states and, conversely, how BLEC function may in turn modulate sleep.