Formation of urea from ammonium
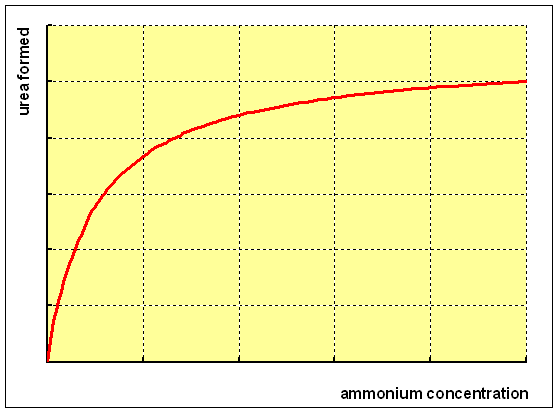 If
you incubate isolated hepatocytes with increasing concentrations of ammonium,
you will see that there is a steady increase in the formation of urea at low
concentrations of ammonium, levelling off as the pathway for urea formation
becomes saturated. If you perform such studies with isotopically labelled ammonium
(15N, a stable isotope) you will find that one of the two N atoms in urea comes
from ammonium, but the other does not - it arises from the amino group of the
amino acid aspartate.
If
you incubate isolated hepatocytes with increasing concentrations of ammonium,
you will see that there is a steady increase in the formation of urea at low
concentrations of ammonium, levelling off as the pathway for urea formation
becomes saturated. If you perform such studies with isotopically labelled ammonium
(15N, a stable isotope) you will find that one of the two N atoms in urea comes
from ammonium, but the other does not - it arises from the amino group of the
amino acid aspartate.
This is essentially the control for the experiments you will perform in the
simulation exercise, when you will look at the effects of adding various compounds,
with and without ammonium ions also present, on the formation of urea.
The sources of ammonium ions for urea synthesis
It is obvious from the discussion of ammonia toxicity that
there is little or no free ammonium in the bloodstream, nor can the intracellular
concentration of ammonium be allowed to rise significantly. Therefore there
has to be a way of forming ammonium as it is required.
There are two ways in which ammonium is formed in the liver: from glutamine
by the action of glutaminase, and from adenosine, by the action of adenosine
deaminase. Each provides about half the ammonium that is incorporated into urea
directly.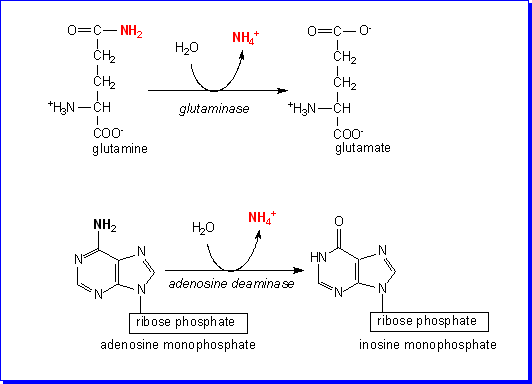
The utilisation of ammonium ions
Ammonium is formed, more or less as required, by the actions of glutaminase
and adenosine deaminase, and is then rapidly used for the formation of carbamyl
phosphate, so that there is little or no accumulation of free ammonium in the
cell. 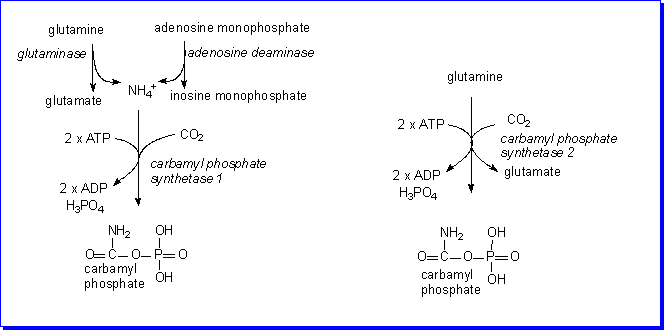
There are two isoenzymes of carbamyl phosphate synthetase in liver cells:
carbamyl phosphate synthetase 1 is a mitochondrial enzyme;
it uses ammonium as the source of nitrogen, and is induced by feeding a high-protein
diet.
carbamyl phosphate synthetase 2 is a cytosolic enzyme; it
uses glutamine directly as the source of nitrogen, and is inhibited by pyrimidine
nucleotides. (Carbamyl phosphate is the starting substrate for pyrimidine synthesis).
Which isoenzyme is likely to be important for
the synthesis of urea?
 If
you incubate isolated hepatocytes with increasing concentrations of ammonium,
you will see that there is a steady increase in the formation of urea at low
concentrations of ammonium, levelling off as the pathway for urea formation
becomes saturated. If you perform such studies with isotopically labelled ammonium
(15N, a stable isotope) you will find that one of the two N atoms in urea comes
from ammonium, but the other does not - it arises from the amino group of the
amino acid aspartate.
If
you incubate isolated hepatocytes with increasing concentrations of ammonium,
you will see that there is a steady increase in the formation of urea at low
concentrations of ammonium, levelling off as the pathway for urea formation
becomes saturated. If you perform such studies with isotopically labelled ammonium
(15N, a stable isotope) you will find that one of the two N atoms in urea comes
from ammonium, but the other does not - it arises from the amino group of the
amino acid aspartate.
