Total amino acid composition of the peptide
The first step in determining the amino acid sequence of a peptide is to find
out which amino acids are present, and how many of each.
This is achieved by hydrolysing the peptide, then subjecting the hydrolysate
to high pressure liquid chromatography to identify (and quantify) the amino
acids present.
Peptide hydrolysis and sample preparation
The peptide is subjected to complete acid hydrolysis by heating overnight at
105C in 10 mol/L hydrochloric acid, in a sealed tube. This hydrolyses all peptide
bonds, releasing free amino acids. However, it also results in destruction of
tryptophan, so if you suspect that your peptide contains tryptophan (e.g. from
its absorption spectrum) then you may need to use alkaline or enzymic hydrolysis.
The hydrolysate is then neutralised, and reacted with o-phthaldialdehyde and
mercaptoethanol to form fluorescent iso-indole derivatives, before being injected
onto an hplc column.
High-pressure liquid chromatography
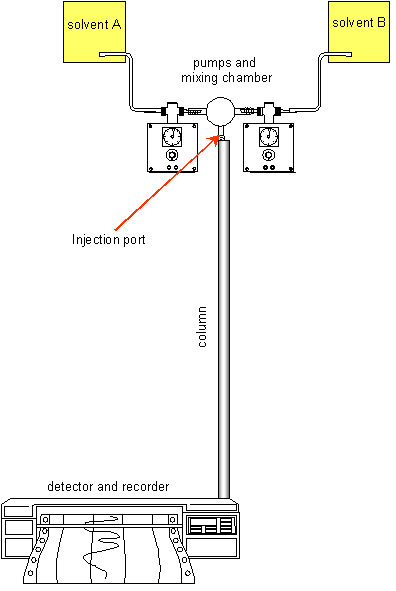 Chromatography
depends on the partition of solutes between a stationary phase (usually a solid)
and a mobile phase (usually a liquid, but sometimes a gas). Solutes that are
more tightly adsorbed onto the stationary phase travel more slowly, and therefore
in column chromatography are eluted from the column later than solutes that
are less tightly adsorbed by the stationary phase. The time at which a compound
is eluted from the column is its retention time. The separation of different
solutes depends on their chemistry, the nature of the stationary phase and the
composition of the mobile phase.
Chromatography
depends on the partition of solutes between a stationary phase (usually a solid)
and a mobile phase (usually a liquid, but sometimes a gas). Solutes that are
more tightly adsorbed onto the stationary phase travel more slowly, and therefore
in column chromatography are eluted from the column later than solutes that
are less tightly adsorbed by the stationary phase. The time at which a compound
is eluted from the column is its retention time. The separation of different
solutes depends on their chemistry, the nature of the stationary phase and the
composition of the mobile phase.
High pressure liquid chromatography (hplc) is an extremely sensitive column
method, using very small volumes of sample, and columns that are typically some
150 mm long and 4 - 5 mm in diameter. The stationary phase is in the form of
very uniform spheres, typically some 5 µm in diameter, packed into the
column. The mobile phase is pumped through the column under high pressure, and
5 - 10 µL of the sample is injected into the system just above the column.
Sometimes the mobile phase has a constant composition; at other times its composition
is varied though the elution period by mixing two different solutions to provide
a gradient of mobile phase composition.
In some hplc systems the sample is derivatised before chromatography (as in
this case, where it is the OPT-derivatives of the amino acids that are subjected
to hplc); in other cases the eluate is derivatised (post-column derivatisation)
to form coloured or fluorescent derivatives. In some cases the detection system
is such that no derivatisation is required before or after chromatography.
The eluate then passes through a detection system, which may be electrochemical,
or may rely on absorption of light, of fluorescence. In the case case of OPT-derivatives
of amino acids, the detection system is fluorimetric.
The hplc that is used in this simulation uses a reverse phase (C18) column,
with a 12.5 mmol/L sodium phosphate buffer at pH 7.2, and a gradient of 10 -
50% acetonitrile. The eluate is monitored using a fluorescence detector with
excitation at 340 nm and emission at 445 nm.
The figure below shows typical traces for a mixture of standard amino acids
(left) and a peptide hydrolysate (right), so that it is possible to identify
which amino acids are present in the peptide.

Fluorescence versus absorption spectrophotometry
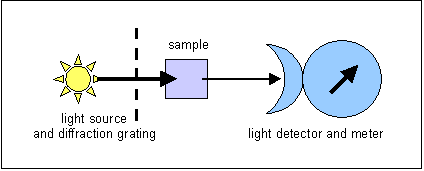 In absorption
spectrophotometry, light of s defined wavelength is shone through the sample,
and some is absorbed.The absorption of light is determined by the concentration
of solute and the path length. What is measured is the light that is transmitted
through the solution. This means that at low concentrations of solute, sensitivity
is low, since there is little difference between the intensity of the incident
light (that shone into the sample) and that of the transmitted light, because
little has been absorbed.
In absorption
spectrophotometry, light of s defined wavelength is shone through the sample,
and some is absorbed.The absorption of light is determined by the concentration
of solute and the path length. What is measured is the light that is transmitted
through the solution. This means that at low concentrations of solute, sensitivity
is low, since there is little difference between the intensity of the incident
light (that shone into the sample) and that of the transmitted light, because
little has been absorbed.
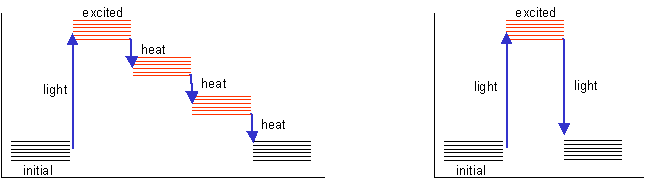
The light energy is used to excite electrons in the solute (this is why the
wavelength is more or less specific for the compound under consideration). These
electrons normally return to the resting state in a series of small jumps, losing
the energy of excitation as heat, as shown on the left in the diagram below.
In some compounds, the energy of excitation is lost in a single quantum jump,
as light, as shown on the right in the diagram below. Such compounds are said
to be fluorescent - if they are excited with light of an appropriate wavelength,
they will emit light of a longer wavelength (lower energy).
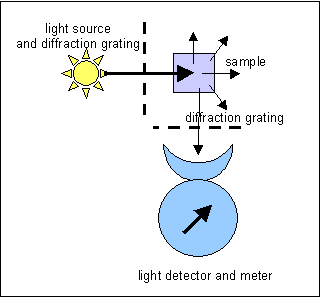 The
light is emitted in all directions, and in a spectrophotofluorimeter what is
measured is the light emitted at right angles to the incident light. This permits
considerably greater sensitivity than absorption spectrophotometry, since, rather
than looking for a small diminution in the intensity of the incident light,
we are measuring light emitted without interference from the transmitted light.
At least in theory, photomultipliers can detect a single photon.
The
light is emitted in all directions, and in a spectrophotofluorimeter what is
measured is the light emitted at right angles to the incident light. This permits
considerably greater sensitivity than absorption spectrophotometry, since, rather
than looking for a small diminution in the intensity of the incident light,
we are measuring light emitted without interference from the transmitted light.
At least in theory, photomultipliers can detect a single photon.
Spectrophotofluorimetry also permits a higher degree of specificity than absorption
spectrophotometry, since diffraction gratings can be used to select not only
the wavelength of the incident light, but also that of the emitted light.
 Chromatography
depends on the partition of solutes between a stationary phase (usually a solid)
and a mobile phase (usually a liquid, but sometimes a gas). Solutes that are
more tightly adsorbed onto the stationary phase travel more slowly, and therefore
in column chromatography are eluted from the column later than solutes that
are less tightly adsorbed by the stationary phase. The time at which a compound
is eluted from the column is its retention time. The separation of different
solutes depends on their chemistry, the nature of the stationary phase and the
composition of the mobile phase.
Chromatography
depends on the partition of solutes between a stationary phase (usually a solid)
and a mobile phase (usually a liquid, but sometimes a gas). Solutes that are
more tightly adsorbed onto the stationary phase travel more slowly, and therefore
in column chromatography are eluted from the column later than solutes that
are less tightly adsorbed by the stationary phase. The time at which a compound
is eluted from the column is its retention time. The separation of different
solutes depends on their chemistry, the nature of the stationary phase and the
composition of the mobile phase.
 In absorption
spectrophotometry, light of s defined wavelength is shone through the sample,
and some is absorbed.The absorption of light is determined by the concentration
of solute and the path length. What is measured is the light that is transmitted
through the solution. This means that at low concentrations of solute, sensitivity
is low, since there is little difference between the intensity of the incident
light (that shone into the sample) and that of the transmitted light, because
little has been absorbed.
In absorption
spectrophotometry, light of s defined wavelength is shone through the sample,
and some is absorbed.The absorption of light is determined by the concentration
of solute and the path length. What is measured is the light that is transmitted
through the solution. This means that at low concentrations of solute, sensitivity
is low, since there is little difference between the intensity of the incident
light (that shone into the sample) and that of the transmitted light, because
little has been absorbed.
 The
light is emitted in all directions, and in a spectrophotofluorimeter what is
measured is the light emitted at right angles to the incident light. This permits
considerably greater sensitivity than absorption spectrophotometry, since, rather
than looking for a small diminution in the intensity of the incident light,
we are measuring light emitted without interference from the transmitted light.
At least in theory, photomultipliers can detect a single photon.
The
light is emitted in all directions, and in a spectrophotofluorimeter what is
measured is the light emitted at right angles to the incident light. This permits
considerably greater sensitivity than absorption spectrophotometry, since, rather
than looking for a small diminution in the intensity of the incident light,
we are measuring light emitted without interference from the transmitted light.
At least in theory, photomultipliers can detect a single photon.