There are 8 antisera available. They have been raised in rabbits that were immunised with oestradiol covalently bound to serum albumin as the immunogen. Each animal will respond differently to the immunogen, and as a result some antisera will have a high titre (i.e. a high concentration of anti-oestrogen antibodies) and some will have a low titre. Obviously, it is desirable to have as high an antibody titre as possible, since this minimises the amount of antiserum that you have to use for each assay.
The antibody titre is defined as the dilution of the antiserum that will bind 50% of the radioactivity when incubated with tracer alone (i.e. with no additional non-radioactive oestradiol). To determine the titre, you will incubate a range of dilutions of the antiserum with tracer, and plot the percentage bound against the dilution of the antiserum (on a semilogarithmic scale).
For future studies you should use a dilution of the antiserum that gives approximately 50% binding of the tracer, since this gives the best sensitivity for the assay.
Some of the antisera will contain proteins that bind the some of the tracer radioactive oestradiol non-specifically; this is useless if the aim is to use specific oestradiol binding as a means of measuring oestradiol in samples. It is easy to determine whether the binding is specific (i.e. to oestradiol binding sites) or non-specific (i.e. to sites that have a general affinity for lipophilic molecules), since if the binding is specific then the bound radioactive ligand will be displaced by an increasing concentration of oestradiol, while non-specifically bound radioactivity will not.
In the studies of antiserum dilution to determine the antibody titre of each antiserum, you will also carry out a series of incubations in the presence of a saturating amount (10 nmol) of non-radioactive oestradiol as well as the radioactive tracer. This should displace more or less all of the bound radioactivity if there is not much non-specific binding.
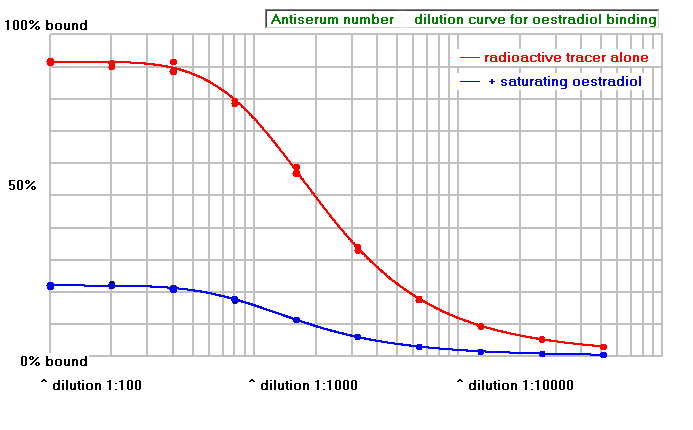
A good antiserum that is likely to be useful for radio-immunoassay will have a high titre, and low non-specific binding, as shown in the graph below. In this case the antibody titre (the dilution that gives 50% binding of tracer ligand) is around a dilution of 1:1000 (there are points at a dilution of 1:800 and 1:1600)

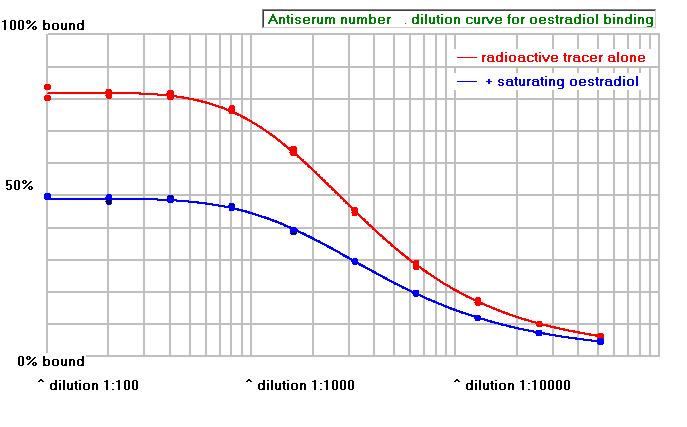
The graph below shows an antiserum that will not be useful. Although again it has an antibody titre around 1:1000, little of the radioactive tracer is displaced by the saturating non-radioactive oestradiol, showing that there is a great deal of non-specific binding.
For each of the antisera you will determine the percentage of radioactive oestradiol that is bound to protein as the antiserum is diluted over a range from 1:100 to 1:102,400.
All solutions are prepared in, and diluted with, phosphate buffered saline (pH 7.4) containing 1 g/L bovine serum albumin (PBS+BSA). The incubations at 4C overnight are prepared as:
100 µL tritiated oestradiol (50,000 dpm per assay)
100 µL PBS+BSA or 10 nmol oestradiol in 100 µL PBS+BSA
100 µL diluted antiserum
At the end of the incubation 200 µL of charcoal suspension in PBS+BSA is added and the mixture is centrifuged.
200 µL aliquots of the supernatant (= bound ligand) are used for liquid scintillation counting