| home |
| resume |
| research |
| publications |
| teaching |
| contact |
| Prior Research Work |
During my PhD studies at the University of California San Diego / San Diego State University I extensively studied graphite and graphene-based materials and their chemistry with common atmospheric molecules such as molecular oxygen, water and carbon monoxide. Furthermore, these materials have unique electronic, mechanical and magnetoelectronic properties making them excellent candidates in applications such as solar harvesting and nanoelectro-mechanical devices. Currently, as a postdoctoral researcher at the University of Cyprus, I study the mechanism and kinetics of catalytic reactions occurring on nanoparticles and nanocatalysts that find industrial applications, such as the water-gas shift (WGS) for the production of high-purity H2 from CO obtained from renewable energy sources (i.e. biomass, bio-ethanol, bio-oil). Overall, I have been able to expand on my knowledge in the general field of Physical Chemistry using both experimental and theoretical approaches and by taking part in a broad range of scientific projects most of which are highlighted in the following sections. |
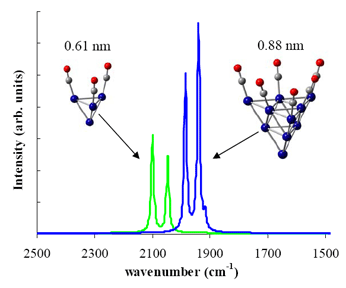
Trends of CO adsorption on transition and noble metal nanoclusters1-3 |
Many commercial and industrial catalysts promote the reaction rate of chemical processes that involve the adsorption of CO on supported-metal catalysts. Examples include the Fischer-Tropsch reaction for the production of liquid fuels or the WGS reaction used to produce fuel-cell-compatible hydrogen. Furthermore, CO is highly toxic to living organisms and is one of the major atmospheric pollutants. In this study we used Diffuse Reflectance Fourier Transform Spectroscopy (DRIFTS) to obtain in situ spectra under reaction conditions and Density Functional Theory (DFT) computations to carry out the spectral assignment. The trends of CO adsorption on various transition metals derived in this study are important for the rationalization of catalytic activities of reactions that involve the adsorption of CO. |
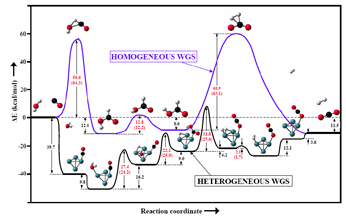
Water-promoted reaction mechanism and kinetics of the WGS reaction4-5 |
We elucidate the mechanism of the heterogeneous and homogeneous WGS reaction. The rate determining steps were assigned to water dissociation and formate association and formate decomposition. A water-mediated mechanism is proposed for the first time in which favorable H-bonding interactions stabilize Zundel-cations adsorbed to the metal cluster, leading to a lower energy path for the dissociation of water. These findings suggest that operation of heterogeneous WGS catalysts in a cyclic fashion where water is first pre-adsorbed might enhance the catalyst performance and CO conversion turnover frequencies. |
A new interpretation of the STM image of graphite6-8 |
Highly-oriented-pyrolytic-graphite (HOPG), a synthetic form of graphite, has been used as a standard for STM calibration for over two decades. The majority of the STM images of graphite show only three of the six carbon atoms in a given six-member ring. Several theories emerged to explain these interesting features such as the carbon site structural asymmetry and tip effects. Through Scanning Tunneling Microscopy (STM) experiments we obtained highly-resolved STM images of graphite basal plane and performed theoretical computations in an attempt to explain the mystery that surrounds the image. Our findings indicate that what we see corresponds to the electron density of π-states, a rare case where the wavefunctions of a material can be directly observed.
|
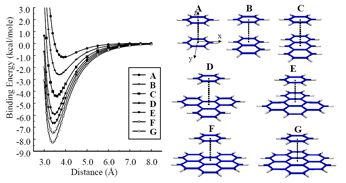
Study of π-π interactions and molecular polarizability in PAHs and polyynes9-11 |
Dispersion interactions are an important driving force for the supermolecular complexation and dynamics of Polycyclic Aromatic Hydrocarbons (PAHs). PAHs are the major ingredient of gas-phase soots a product from the incomplete combustion of hydrocarbons. This study aimed to derive semiquantitative relationships that can be used to estimate the binding energy, equilibrium separation and potential energy surface (PES) for supermolecules consisting of PAHs. An interesting linear correlation between the binding energy and the polarizability is derived that makes it possible to predict these interactions on the basis of molecular polarizabilities.These findings prompted us to extend our work to polyynes, a promising material for nonlinear optical (NLO) applications, and derive a quantitative structure-property relationship, which can estimate the static longitudinal and transverse polarizability in polyynes as a function of their length (L). The decrease of the polarizability length dependence is shown to predominantly arise from electron-electron repulsion rather than electron correlation. Decrease of the electron-electron repulsion term is suggested to be the key term in enhancing NLO characteristics of linear polymeric materials.
|
Ion induced defects on graphite using STM and differential reflectance spectroscopy12-14 |
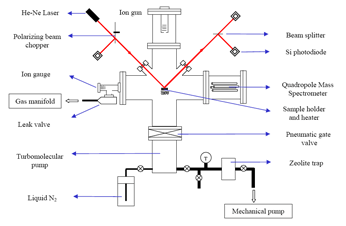
The extreme thermal conductivity, chemical inertness and thermochemical properties of HOPG make it a suitable material for many high-temperature applications. Examples include its use as protective material in spacecrafts and in inner walls of fusion reactors. However, the extreme conditions under which a spacecraft is exposed and the working conditions of fusion reactors cause strong erosion of graphite due to ion bombardment. In this study we have designed and built an Ultra-high vacuum chamber which was used to study the ion-induced damages on HOPG. We determine the threshold energy for ion penetration and extent of surface damage under inert and oxidative conditions using STM and optical differential reflectance spectroscopy. |
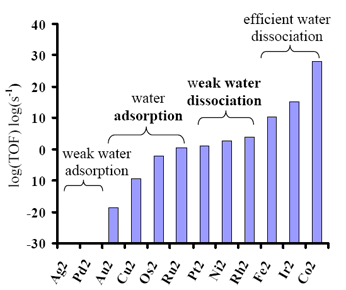
Kinetics of water dissociation on metal nanoclusters 15-16 |
Finding materials that have enhanced activity towards the dissociation of water is of importance for the production of molecular hydrogen (H2) from water and for the rationalization of the reaction kinetics of humidity with atmospheric particulate matter. Water is abundant in nature and the fact that the oxidation of H2 yields water means that it can be infinitely recycled with no major waste products, thus claimed to be the most environmentally friendly fuel currently known. In this study, we performed a systematic analysis of the adsorption and dissociation of water on transition and noble metal dimers. DFT simulations indicated that the dissociation of water on these clusters is thermally driven even in the absence of other electrocatalytically and photocatalytically driven processes. Two reaction pathways were found with turn-over-frequencies (TOFs) for water dissociation given by the following series Co2 > Ir2 > Fe2 > Rh2 > Ni2 > Pt2 > Ru2 > Os2 > Cu2 > Au2 > Pd2 > Ag2 at standard ambient temperature and pressure. Linear-free energy relationships were derived, that can predict the dissociation barrier of water on transition and noble metals as a function of free energy change for dissociation. The calculated TOFs correlate well with experimental evidence from Temperature Programmed Desorption Spectroscopy (TPDS), after water adsorption on evaporated Fe, Co, and Ni films. In the future I plan to elucidate the complete pathway for the production of H2 from H2O on metals such as Co and Ir that have exhibited high activity towards the dissociation of water. |
Size-dependent elastic properties of Graphene Nanoribbons17 |
Graphene Nanoribbons (GNRs) have been suggested as the most sensitive material for use as nanoelectromechanical systems (NEMS) for force, mass and charge detection. Therefore, the accurate determination of the size-dependent elastic properties of GNRs are desirable for fundamental engineering applications. In a preliminary study we determine the size-dependent in-plane Young’s, shear and bending moduli and Poisson’s ratio of the homologous GNR series (n = 2-12) with the use of accurate all electron first principle computations. In the future we want to assess the effect of chemical species bound at the periphery of the GNRs to the mechanical and vibrational properties of these materials. |
|
References |
|
| Figure 1. Simulated IR absorption spectra of adsorbed CO on Rh nanoclusters. | ||
|
||
| Figure 2. PES of the kinetic path of the homogeneous and heterogeneous WGS reaction. | ||
 |
||
Figure 3. Experimental (top) and simulated (bottom) STM images of graphite. |
||
 |
||
Figure 4. PES of benzene interacting with various PAHs. |
||
 |
||
Figure 5. Simplified schematic of home-made experimental setup |
||
 |
||
Figure 6. Bar diagram of TOF logarithm for water dissociation on transition and noble metal dimers. |
||
Copyright 2015 Constantinos Zeinalipour
Webpage page was last updated on: 15 December, 2009 legal